Shutterstock/Megan Betteridge
Take a stroll through any supermarket, and you’ll find a variety of fermented products. Wine and beer are fermented from fruits and grains, respectively. Yogurt, sour cream, and some cheeses are fermented dairy products. Chocolate is made from fermented cacao beans. Sauerkraut and kimchi are fermented cabbage. The little bubbles inside leavened bread are the result of fermentation. So is the slight tang in sourdough. The acetic acid in vinegar comes from fermentation. And fermentation isn’t just limited to the foods we eat; it takes place all around us. Odds are good that the gas in your car contains ethanol — the product of industrial fermentation. Even your own muscle cells will start to ferment if you work them hard enough.
People who ferment foods or run industrial fermentations often loosely define fermentation as a process in which microorganisms — such as yeast or bacteria — break down organic matter in a sealed container. This leaves out fermentations that occur in the wild and in multicellular organisms — especially the muscles of vertebrates — but covers the fermentations intentionally performed by humans.
Adobe Stock/Yulia Furman
Biologically, fermentation is a metabolic process in which a carbohydrate is broken down anaerobically, resulting in the release of energy. In layman’s terms, fermentation is a process that occurs inside living organisms, in which molecules that contain both carbon and hydrogen (such as sugars) are broken down in an environment that lacks oxygen. As a result, energy that the organism can use is released. This is a complex process, and I’ve discussed the molecular details first. If you’d rather get the conceptual overview first, scroll down to “Why Do Some Organisms Ferment?”, and then come back. In some food fermentations, the most important actions of the microorganisms are not biological fermentations at all. For example, the breakdown of proteins and fats in fermented fish may contribute more to the desired qualities of the food than the breakdown of carbohydrates does. Sometimes this process is called “ripening” instead of “fermentation.”
Fermentation is one of the many interrelated metabolic pathways that keep life going on Earth. The best introduction to fermentation, I think, is to see how it fits into that big metabolic picture. Fermentation requires carbohydrates — often the sugar glucose — and is an alternative to aerobic cellular respiration in many organisms. We’ll take a quick look at photosynthesis, which produces glucose, and at cellular respiration before getting into the nitty-gritty of fermentation.
It All Starts with Photosynthesis
Life requires energy, and that energy ultimately comes from the sun. Plants, algae and other protists, and some types of bacteria harness the sun’s energy directly through the process of photosynthesis. In photosynthesis, the energy in light is used to join water (H2O) and carbon dioxide gas (CO2) to produce glucose (C6H12O6) and oxygen gas (O2). This happens in a series of steps, but the net reaction is:
Glucose, a sugar, becomes an energy source for the organisms that produce it and for those that consume those organisms — animals, fungi, some unicellular nonbacteria, a very few nonphotosynthetic plants, and many bacteria. Animals can obtain glucose either from eating photosynthetic organisms, or by eating other animals that have eaten plants.
The energy in glucose can be accessed through either aerobic respiration or fermentation, but much more energy is released via respiration.
Before I continue to glycolysis — the initial step in the glucose breakdown process of both fermentation and aerobic respiration — you might find it helpful to know more about aerobic respiration and how it relates to fermentation.
Aerobic Respiration
Respiration occurs in animals, plants, fungi, most protists, and many bacteria. In the case of aerobic respiration — or respiration that uses oxygen — the net reaction is:
Notice that this is almost the exact opposite of the net reaction of photosynthesis. The only difference is that light energy functions as a reactant in photosynthesis, while chemical energy and heat are products of aerobic respiration. The chemical energy is in the form of adenosine triphosphate (ATP), a molecule that stores and releases energy in cells. In theory, 38 molecules of ATP could be generated from every molecule of glucose. (In practice, this isn’t achieved for a variety of reasons.) By contrast, fermentation theoretically yields two ATP per molecule of glucose.
One of the reasons aerobic respiration is so much more efficient than fermentation is that oxygen is employed as an outside electron receptor. To make a long story short — as this is an article on fermentation, not aerobic respiration — oxygen is a very effective electron acceptor. During aerobic respiration, electrons are transferred from electron donor molecules, often NADH or FADH2, through a series of electron acceptors (the electron transport chain), to oxygen. Enough energy is generated through this process to synthesize ATP molecules by adding phosphate groups to adenosine diphosphate (ADP) molecules.
Glycolysis
Aerobic respiration and fermentation share an initial pathway, called “glycolysis,” in which one glucose molecule is converted into two pyruvate molecules. The figure at right shows the path of the six carbon atoms in a glucose molecule as it’s changed into two three-carbon pyruvate molecules.
The first several steps of glycolysis — leading from one molecule of glucose to two molecules of glyceraldehyde-3-phosphate — are sometimes called the “preparatory phase.” Two ATP are converted into ADP at this stage; in other words, energy is consumed, not released. In addition, two NAD+ molecules accept electrons to form two NADH molecules — chemically, they’ve been reduced.
The second phase, sometimes called the “payoff phase,” converts the two molecules of glyceraldehyde-3-phosphate into molecules of pyruvate. This conversion forms four ATP molecules: a net ATP gain of two. Each of the two NADH formed in the first phase can yield three ATP through the electron transport chain if oxygen is present, and the two pyruvate molecules can each yield 15 ATP through the citric acid cycle.
The Citric Acid Cycle
Here’s where the fermentation and aerobic respiration pathways part ways. I’ll skip the in-depth details of the citric acid cycle — a 10-step cycle of reactions that completes the respiration pathway — and just give the outcome. In respiration, pyruvate is next converted to a molecule called “acetyl-CoA.” This in turn is converted into citric acid, the molecule the cycle is named for. As it passes through the cycle, each citric acid molecule forms one molecule of guanosine triphosphate (GTP), four molecules of NADH, and one molecule of FADH2. (GTP is a molecule similar to ATP and with an equivalent energy reserve. FADH2 is similar to NADH.) The GTP is then converted to ATP, and the NADH and FADH2 molecules deliver electrons through the electron transport chain that eventually end up being accepted by oxygen. This produces 15 ATP per pyruvate molecule, or 30 ATP per original glucose molecule. Remember, each molecule of glucose yielded two ATP and two NADH through glycolysis as well (eight total ATP from glycolysis, after those two NADH pass through the electron transport chain). Aerobic respiration yields a theoretical total of 38 ATP per molecule of glucose. At that point, all the carbon molecules from the initial glucose molecule have been incorporated into CO2 molecules, and the NADH molecules that donated their electrons to the electron transport chain have reverted to NAD+.
Anaerobic Respiration
In some bacteria, respiration can occur anaerobically (without air) as well. In anaerobic respiration, glucose breakdown follows the same path — through glycolysis and then the citric acid cycle — but the final electron acceptor is something other than oxygen. Iron, manganese, cobalt, uranium, nitrate, fumarate, sulfate, and other molecules can be electron acceptors, though each item in the list is progressively worse at accepting electrons. Iron, for example, isn’t as good an electron acceptor as oxygen, and manganese is still less effective. Less energy is produced when these molecules serve as final electron acceptors, but there are still organisms that function by anaerobically respiring with them.
Take a Breath
So far we’ve learned: Both fermentation and respiration start with glucose and follow the glycolytic pathway. Glycolysis yields two pyruvate molecules, two ATP, and two NADH per glucose molecule. If the pyruvate gets fed into the citric acid cycle, and the NADH into the electron transport chain, 38 ATP are possible under aerobic conditions. A key point to understand here, before we tackle fermentation, is that the two pyruvate molecules contain almost as much energy as the initial molecule of glucose — most of the stored energy is released in the citric acid cycle, not in glycolysis. Most of the energy released in the citric acid cycle comes from NADH molecules donating electrons to the electron transport chain. In anaerobic respiration, NADH and FADH2 donate electrons to an electron transport chain with a lower-affinity electron acceptor than oxygen. Less ATP gets produced this way, but still more than the two produced via glycolysis.
Finally, Fermentation
There are two main types of fermentation: ethanol and lactate. Both types of fermentation, like aerobic respiration, start with glycolysis. In ethanol fermentation, there are two more steps; in lactate fermentation, only one.
Ethanol Fermentation
In ethanol fermentation, after glycolysis, pyruvate is converted to acetaldehyde, and then acetaldehyde to ethanol. The enzyme pyruvate decarboxylase catalyzes the first reaction, consuming one proton (H+) and releasing one molecule of CO2 per pyruvate. Alcohol dehydrogenase catalyzes the second reaction, using one NADH molecule and one proton (H+) per acetaldehyde. The two NADH produced in glycolysis are thus both returned to NAD+. The net yield is just the two ATP produced during glycolysis.
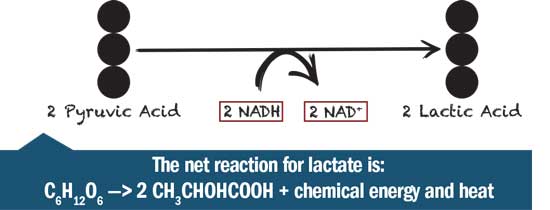
Lactate Fermentation
In lactate fermentation, after glycolysis, pyruvate is converted to lactate (or lactic acid) by the enzyme lactate dehydrogenase. This also converts an NADH back to NAD+. No CO2 or other gas is released, and again the net yield is two ATP.
Summary of Fermentation
In both of the common types of fermentation, one glucose molecule is broken down into two pyruvate molecules via glycolysis. Pyruvate is then reduced to form either ethanol or lactate. No new ATP molecules are produced after glycolysis. In addition, the NADH molecules — which produce ATP via the electron transport chain in respiration — give up their electrons to form NAD+. So, the net energy gain from fermentation — two molecules of ATP — is far less than would have been gained through respiration. That energy still exists, locked in the end products of fermentation — ethanol and lactate (or lactic acid, the conjugate acid of lactate). In contrast, the end products of aerobic respiration — CO2 and H2O — can’t be broken down by an organism to yield more energy.
Why Do Some Organisms Ferment?
Given that respiration — either aerobic or anaerobic — yields so much more energy, it’s reasonable to ask why any organisms ferment at all. Why not just respire all the time, getting more energy per glucose molecule consumed?
Many yeast and bacteria can actually respire when oxygen is present and switch to fermentation when it isn’t. Some organisms stick to fermentation because they live in permanently anoxic environments. Because they never encounter oxygen, they can’t respire; in fact, oxygen is toxic to some anaerobic organisms.
Further, not all organisms have evolved or acquired the ability to respire. Respiration occurs in only a few branches of bacteria, and in eukaryotic organisms — those with “true” nuclei. Glycolysis occurs in the cytoplasm of eukaryotic organisms, whereas the citric acid cycle occurs in mitochondria. Why the separation?
The mitochondria in eukaryotic cells were once free-living bacterial cells with the ability to respire, which either colonized or were engulfed by ancestral eukaryotic cells. Mitochondria still have the remnants of their bacterial genome and reproduce independently inside the cell. Some organisms that don’t respire are simply descended from ancestors that were never colonized by a respiring bacteria.
GettyImages/DejanKolar
An odd phenomenon in some yeasts, called the “Crabtree Effect,” hints at fermentation having benefits that outweigh the loss in ATP production efficiency. In an environment that contains both oxygen and a high concentration of sugar, yeast will ferment rather than respire. Why lose out on the energetic benefits of respiration? The answer may be that alcohol is a poison to most microorganisms; many cannot survive when the alcohol concentration of the environment exceeds a few percent. In contrast, some yeasts can survive alcohol concentrations in the high teens. When sugar is plentiful, yeasts ferment — likely because this poisons their competitors and allows them to monopolize the sugar source.
Some fermenting microorganisms only perform one type of fermentation. These are called homofermentative organisms. Saccharomyces, the yeast that ferments beer and wine, only performs ethanol fermentations. Other organisms can produce both ethanol and lactate; these are called heterofermentative organisms. For example, bacteria in the genus Leuconostoc produce both ethanol and lactate. Lactobacillus, a bacterial genus seen in many food fermentations, can be either homofermentative or heterofermentative, depending on the species.
The two most common types of fermentation start with glucose and end with the production of ethanol or lactate, but there are other methods. Other sugars can be fermented, often by first being converted to glucose or fructose, which is one of the steps in glycolysis. Interestingly, the end products of ethanol and lactate fermentation can themselves be fermented by some organisms.
Ready for a Drink?
In humans, alcohol enters the bloodstream and travels to the liver. There, it’s converted to acetaldehyde by alcohol dehydrogenase, the same enzyme that catalyzed the reverse action in the fermenting organism. Acetaldehyde is then converted to acetyl acid, and then to acetyl-CoA, which enters the citric acid cycle and produces ATP.
Likewise, lactate gets transferred to the human liver, where it’s converted back into glucose through a metabolic pathway called “gluconeogenesis.” This pathway requires an input of energy (from ATP), but the energy in the lactate is mostly preserved.
A practical consequence of fermentation leaving behind molecules that can be further broken down to yield energy is that fermented foods and beverages retain most of the ingredients’ original caloric load.
The Big Picture
So that’s fermentation, framed by two major metabolic pathways connected to it. Photosynthesis harnesses the sun’s energy to turn carbon dioxide and water into glucose. Respiration does the opposite, breaking sugar down to pyruvate (via glycolysis) and then into water and carbon dioxide. This releases all of the usable energy stored in the sugar molecule. Fermentation also breaks down sugar into pyruvate, but only releases a small amount of the sugar’s stored energy, preserving most of that energy in the form of ethanol or lactate. Humans have exploited fermentation for both ethanol production and food preservation, not to mention for the intriguing flavors that result from fermentation.
Chris Colby is on the Fermentation advisory board, and is the author of two books, Home Brew Recipe Bible and Methods of Modern Homebrewing (Page Street Publishing), and over 100 articles on brewing beer, appearing in such publications as Brew Your Own and Zymurgy. He’s currently a contributing editor to Beer and Wine Journal. He lives with his wife and their cats in Bastrop, Texas. Find him on Twitter @ColbyBrew.